Sumayah Askandarani¹, Khadija M Alshehabi²*, Somaya Ahmed Aleissa¹, Abdulaziz Saleh Alghamdi¹, Baher Mohamed Algadaa¹, Fahad E Alotaibe¹, Abdulnaser Alabadi¹
1Multi-Organ Transplant Center, King Fahad Specialist Hospital, Eastern Health Cluster, Saudi Arabia
2Department of Nephrology, Salmaniya Medical Complex, Government Hospitals, Bahrain
*Corresponding Author: Khadija M Alshehabi, Department of Nephrology, Salmaniya Medical Complex, Government Hospitals, Bahrain
Abstract
Malaria is an uncommon but potentially severe complication in solid organ transplant (SOT) recipients. Transmission may occur via donor-derived infection, transfusion exposure, or de novo acquisition in endemic areas. Immunosuppression blunts cytokine-mediated febrile responses, often delaying diagnosis and permitting high-burden parasitemia. We report two kidney transplant recipients who developed Plasmodium falciparum and Plasmodium vivax malaria following overseas transplant procedures. These cases highlight diagnostic pitfalls related to afebrile presentation, clinical resemblance to rejection or graft-related complications, and the critical importance of epidemiological screening. Both patients responded favorably to intravenous artesunate followed by appropriate oral therapy. Our findings reinforce malaria as a key differential diagnosis in post-transplant recipients presenting with unexplained anemia, encephalopathy, cytopenias, or graft dysfunction, particularly in the setting of transplant tourism.
Keywords: Malaria; Kidney transplantation; Solid organ transplantation (SOT); Transplant tourism; Donor-derived infection;
Immunosuppression
Introduction
Malaria is a rare but well-recognized complication in solid organ transplant (SOT) recipients [1]. Transmission may occur through donor-derived infection, transfusion-related exposure, or post-transplant acquisition in endemic regions [2, 3]. Although uncommon in high-income countries, malaria remains clinically significant in transplant recipients due to impaired immune regulation, atypical clinical presentation, and an increased risk of severe disease [1, 4, 5]. All major human Plasmodium species—P. falciparum, P. vivax, P. ovale, and P. malariae—have been reported in SOT recipients [1].
Malaria remains a major global infection with more than 200 million cases annually, causing substantial morbidity and mortality, particularly in Africa and South Asia [6, 7]. In immunosuppressed hosts, including transplant recipients, classical febrile patterns may be absent, while severe manifestations such as hemolysis, cerebral involvement, and graft dysfunction occur more frequently [4-8]. These atypical features often delay diagnosis and worsen outcomes.
Transplant tourism—defined as organ transplantation performed outside the recipient’s home country, often within unregulated systems—accounts for an estimated 8–12% of global kidney transplants annually [9-11]. Infectious complications have been reported up to ten-fold more frequently in transplant tourists compared with recipients transplanted in regulated programs [9, 12]. In many non-accredited international centers, donor screening is limited to basic serology, and latent parasitemia remains undetected unless nucleic acid testing is employed [13-15]. Asymptomatic Plasmodium vivax or P. ovale infections may persist for months to years, while P. malariae can remain dormant for decades, enabling transmission from donors without overt symptoms [16-18].
In Saudi Arabia, indigenous malaria transmission has declined substantially and is now largely confined to southern border regions [19, 20]. However, imported malaria remains the predominant burden, particularly among travelers returning from Pakistan, India, Yemen, Sudan, and Ethiopia [21, 22]. Post-transplant patients returning from these regions represent a high-risk subgroup, in whom malaria may present without fever and mimic rejection or anti-thymocyte globulin–related toxicity, further delaying diagnosis and increasing morbidity and mortality [23].
Here, we describe two kidney transplant recipients who developed Plasmodium falciparum and Plasmodium vivax malaria following transplant tourism. We highlight the diagnostic challenges, immunologic masking of symptoms, management considerations, and propose a structured framework for screening returning transplant recipients.
Cases Presentation
Case 1
A 52-year-old man with long-standing hypertension and type 2 diabetes mellitus complicated by retinopathy and nephropathy progressed to end-stage kidney disease (ESKD) and was maintained on peritoneal dialysis. He underwent a living unrelated kidney transplant in Lahore, Pakistan, on July 26, 2025 (transplant tourism). Donor details were unavailable. Induction immunosuppression consisted of three doses of anti-thymocyte globulin (ATG), followed by maintenance therapy with tacrolimus, prednisone, and mycophenolate mofetil.
On July 29, 2025, he presented to our emergency department with an elevated serum creatinine of 530 µmol/L but was clinically stable and afebrile. Renal ultrasonography demonstrated satisfactory graft perfusion with patent vessels. Serological testing was negative for HIV, HBV, and HCV, with evidence of past CMV and EBV infection. An urgent allograft biopsy performed on July 30, 2025 revealed acute cellular rejection (Banff 2024, vascular type 2A) with associated acute tubular injury. He received additional ATG (cumulative dose 5 mg/kg) and was discharged on August 3, 2025 with a creatinine of 292 µmol/L.
During outpatient follow-up, graft function stabilized (baseline creatinine ~200 µmol/L). Maintenance immunosuppression included tacrolimus 2 mg twice daily, tapering prednisone, and mycophenolate mofetil 1000 mg twice daily. Prophylactic therapy included nystatin, valganciclovir, trimethoprim–sulfamethoxazole, and isoniazid with vitamin B6.
On August 12, 2025, he was re-admitted with a three-day history of gastrointestinal symptoms and reduced consciousness. He was pale and disoriented, with tachycardia (108 beats/min) and tachypnea (24 breaths/min), but remained afebrile (37.3 °C). Laboratory results (Table 1) revealed profound anemia (hemoglobin 3.7 g/dL) and thrombocytopenia. At this stage, the differential diagnosis included severe sepsis, acute hemolysis, graft-related complications, ATG-related cytopenia, and viral reactivation; however, the degree of anemia and neurological involvement appeared disproportionate to graft dysfunction alone.
Table 1: The Patient's Initial Laboratory Values
|
Parameter |
Result |
Reference range |
|
Hematology |
|
|
|
Hemoglobin (g/L) |
3.7 |
12–17 |
|
Total white cell count (×10⁹/L) |
21.6 |
4.5–11 |
|
Platelet count (×10⁹/L) |
104 |
150–400 |
|
Biochemistry |
|
|
|
Total bilirubin (µmol/L) |
33 |
5.1–20.5 |
|
Serum albumin (g/L) |
34 |
32–46 |
|
Alanine aminotransferase (U/L) |
19 |
0–55 |
|
Aspartate aminotransferase (U/L) |
12 |
5–34 |
|
C-reactive protein (mg/L) |
205 |
<5 |
|
Lactate (mmol/L) |
3.34 |
<2.0 |
|
Venous blood gas |
|
|
|
pH |
7.26 |
7.32–7.42 |
|
pCO₂ (mmHg) |
22 |
38–50 |
|
HCO₃⁻ (mmol/L) |
12.4 |
22–26 |
|
Renal profile |
|
|
|
Serum creatinine (µmol/L) |
225.2 |
53–97 |
|
Blood urea (mmol/L) |
34 |
3–9.2 |
|
Sodium (mmol/L) |
125 |
136–145 |
|
Potassium (mmol/L) |
7.0 |
3.5–5.1 |
|
Urine examination |
|
|
|
Pus cells |
Negative |
— |
pCO₂, partial pressure of carbon dioxide
He was admitted to the intensive care unit and managed with a massive transfusion protocol. Empiric broad-spectrum antibiotic therapy with meropenem and vancomycin was initiated, and a comprehensive bacterial and viral infectious workup was undertaken. Viral reactivation was considered in view of recent thymoglobulin exposure; however, the absence of organ-specific manifestations and negative virological testing made cytomegalovirus and Epstein–Barr virus disease unlikely. Imaging demonstrated no evidence of active hemorrhage. Given the patient’s recent transplantation in a malaria-endemic region, together with profound anemia, thrombocytopenia, and altered mental status, malaria testing was requested.
On August 13, 2025, the diagnosis of Plasmodium falciparum malaria was definitively established by direct visualization of parasites on a Giemsa-stained thick blood smear. Microscopic examination demonstrated a markedly high parasite burden, with numerous ring-stage parasites graded as 4+, corresponding to more than 10 parasites per microscopic field and an estimated parasitemia exceeding 80,000 parasites per microliter of blood. In addition, a moderate density of gametocytes, graded as 2+, was observed, indicating a substantial presence of sexual-stage parasites.
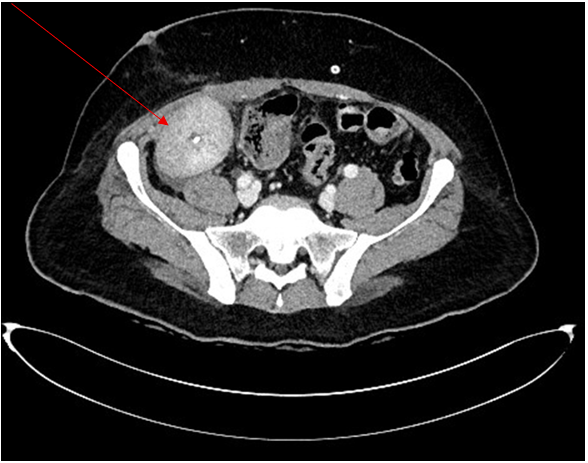
A contrast-enhanced abdominal computed tomography (CT) scan was subsequently performed to assess the renal allograft. Imaging demonstrated heterogeneous peripheral cortical enhancement of the transplanted kidney, findings suggestive of an underlying infectious or inflammatory process. Importantly, no significant abdominal fluid collections or evidence of active bleeding were identified (Figure 1). Concurrent bacterial cultures and virological screening were negative, effectively excluding alternative infectious etiologies.
Intravenous artesunate was initiated at 2.4 mg/kg (210 mg) daily for six days, followed by oral artemether–lumefantrine. Mycophenolate mofetil was reduced to 500 mg twice daily, while tacrolimus levels remained stable. The patient showed rapid clinical and parasitological improvement and was discharged after a 12-day admission with a serum creatinine of 135 µmol/L.
Figure 1: Contrast-enhanced abdominal CT showing heterogeneous peripheral cortical enhancement of the renal allograft, consistent with an inflammatory or infectious process
Case 2
A 42-year-old man with long-standing type 1 diabetes mellitus complicated by retinopathy and nephropathy, and a history of hypertension, progressed to end-stage kidney disease. He underwent a living-unrelated kidney transplant in China in 2016, which failed in 2019 due to chronic rejection. A second living unrelated kidney transplant was performed through transplant tourism on November 29, 2024; no donor information or perioperative medical records were available.
On December 3, 2024, he presented to our emergency department with uremic symptoms and severe graft dysfunction (serum creatinine 1007 µmol/L) but remained afebrile and hemodynamically stable. Doppler ultrasonography demonstrated preserved graft perfusion with elevated arterial velocities at the anastomosis. Urgent hemodialysis was initiated, followed by a renal allograft biopsy, which revealed extensive cortical necrosis and infarction involving 15 of 33 glomeruli. The leading differential diagnoses were acute vascular thrombosis and hyperacute or severe acute rejection. He was treated empirically with anti-thymocyte globulin (total dose 5 mg/kg); however, he remained anuric and dialysis dependent.
Further evaluation with graft ultrasonography, technetium-99m MAG3 renography with SPECT-CT, and CT angiography confirmed irreversible graft thrombosis. Graft nephrectomy was performed on December 9, 2024, with histopathology demonstrating cortical infarction and severe tubular necrosis. By December 13, 2024, the patient was discharged home after discontinuation of mycophenolate mofetil and tacrolimus and was maintained on low-dose corticosteroids alone. Standard antimicrobial prophylaxis was prescribed in view of recent thymoglobulin exposure.
On December 22, 2024, while receiving maintenance dialysis, he developed a one-week history of fever, fatigue, dyspnea, and headache. On admission, he was febrile (38 °C) and tachycardic (117 beats/min) but normotensive. Laboratory findings are summarized in Table 2. Given recent transplantation through transplant tourism, thymoglobulin exposure, and dialysis dependence, the initial differential diagnosis included line-related sepsis, viral reactivation, and delayed complications related to transplant tourism. Empiric broad-spectrum antibiotics were initiated; however, blood cultures, imaging, and virological screening were negative, and no clear bacterial source was identified.
Table 2: The Patient's Initial Laboratory Values
|
Parameter |
Result |
Reference range |
|
Hematology |
|
|
|
Total white cell count (×10⁹/L) |
2.96 |
4.5–11 |
|
Hemoglobin (g/L) |
7.9 |
12–17 |
|
Platelet count (×10⁹/L) |
156 |
150–400 |
|
Reticulocyte count (%) |
2.7 |
0.5–2.5 |
|
Iron studies |
|
|
|
Transferrin saturation (%) |
3 |
20–50 |
|
Hemolysis marker |
|
|
|
Lactate dehydrogenase (U/L) |
407 |
135–225 |
|
Biochemistry |
|
|
|
Liver function tests |
Within normal limits |
— |
|
Renal profile |
|
|
|
Serum creatinine (µmol/L) |
875 |
53–97 |
|
Blood urea (mmol/L) |
17.2 |
3–9.2 |
In view of persistent anemia, recent febrile illness, and epidemiological exposure associated with transplant tourism in a malaria-endemic region, malaria testing was pursued. Peripheral blood smear examination demonstrated Plasmodium vivax trophozoites and gametocytes, with parasitemia estimated at 2%, corresponding to infection of approximately 2% of circulating erythrocytes. These findings established the diagnosis of vivax malaria. On December 23, 2024, intravenous artesunate (2.4 mg/kg; 192 mg) was administered for seven days, resulting in rapid parasite clearance and negative blood smear recorded by December 25, 2024. The patient subsequently received oral primaquine for 14 days to achieve radical cure.
Fever resolved within 48 hours of therapy, with sustained parasitological clearance and clinical improvement. He remained stable on dialysis and was discharged with outpatient follow-up.
Discussion
1. Epidemiology
Malaria remains a major global infectious disease, with an estimated 229–241 million cases reported annually, predominantly in South Asia and Sub-Saharan Africa [6, 7]. Although rare in high-income countries, malaria carries substantial morbidity and mortality in immunocompromised hosts, including solid organ transplant recipients, where severe manifestations are more frequent [1, 4, 5]. In Saudi Arabia, indigenous malaria transmission has declined markedly and is now largely restricted to the southern border regions of Jazan and Najran [19, 20]. However, imported malaria accounts for the majority of cases nationwide, particularly among travelers returning from endemic countries such as Pakistan, Sudan, Yemen, and Ethiopia [21, 22]. Both patients in this report acquired malaria following overseas transplantation in endemic regions, highlighting the ongoing relevance of imported malaria in transplant populations.
2. Risk Factors In Transplant Tourism
Transplant tourism accounts for approximately 8–12% of global kidney transplants and is associated with significantly higher rates of infectious complications compared with regulated transplant programs [9-11]. This increased risk reflects systemic vulnerabilities, including inadequate donor screening, limited access to nucleic acid testing for malaria, and poor traceability of donor medical histories [12-14]. In endemic regions, donors may harbor asymptomatic parasitemia, particularly Plasmodium vivax or P. ovale hypnozoites, or chronic low-grade P. malariae infection, which may persist for years or decades and escape detection by routine microscopy [15-18]. In both cases presented, transplantation in endemic regions without reliable donor documentation substantially increased the likelihood of donor-derived or perioperative malaria acquisition.
3. Pathophysiology In Immunosuppressed Hosts
Immunosuppressive therapy profoundly modifies malaria presentation in transplant recipients. Agents such as anti-thymocyte globulin, tacrolimus, and mycophenolate mofetil impair T-cell–mediated immunity and suppress key pro-inflammatory cytokines, including IL-1, IL-6, IFN-γ, and TNF-α [14, 15]. Consequently, classical febrile responses may be absent despite high parasite burdens, as observed in Case 1. This immunologic blunting permits silent parasite proliferation, increasing the risk of severe anemia, thrombocytopenia, encephalopathy, and graft dysfunction [5, 21, 22]. The overlap between malaria-related organ injury and early rejection further complicates clinical assessment and may delay diagnosis and treatment [14, 15].
4. Diagnostic Challenges
Diagnosing malaria in transplant recipients is challenging because clinical presentation is frequently atypical and may mimic sepsis, acute rejection, drug toxicity, or viral reactivation such as cytomegalovirus or Epstein–Barr virus infection [4, 22, 23]. The absence of fever—particularly in patients receiving potent immunosuppression—can divert clinicians from considering malaria early in the differential diagnosis. Laboratory abnormalities such as anemia and thrombocytopenia are often attributed to other post-transplant complications. Although thick blood smear microscopy remains the diagnostic gold standard, its sensitivity declines significantly at parasite densities below 50 parasites/µL, which are common in asymptomatic or semi-immune individuals [16-18]. Rapid diagnostic tests may aid early detection but cannot reliably quantify parasitemia or identify mixed infections, while molecular assays such as PCR provide superior sensitivity for low-level parasitemia but are not universally available [16-18]. These factors underscore the need for a high index of suspicion in transplant recipients presenting with unexplained cytopenias or neurological symptoms following exposure to endemic regions.
5. Management Considerations
Management of malaria in transplant recipients requires balancing effective parasite eradication with preservation of graft function. Intravenous artesunate is recommended as first-line therapy for severe Plasmodium falciparum infection and was effective in both cases [19-21]. In infections due to P. vivax or P. ovale, primaquine is required to achieve a radical cure by eliminating hypnozoites, following exclusion of glucose-6-phosphate dehydrogenase deficiency [19]. Temporary modification of immunosuppressive regimens—most commonly reduction of mycophenolate mofetil—may be necessary during acute infection, while tacrolimus levels require close monitoring due to potential pharmacokinetic interactions with antimalarial agents and the risk of QT prolongation [22, 23]. Clinicians should also remain vigilant for post-artesunate delayed hemolysis, particularly in patients with underlying renal dysfunction [21].
6. Implications For Saudi Arabia And Global Transplant Programs
These cases highlight important implications for transplant practice at both national and international levels. In Saudi Arabia, where imported malaria predominates despite declining indigenous transmission, structured epidemiological screening should be integrated into post-transplant assessment, particularly for patients who have undergone transplantation abroad [12, 13, 19-22]. At a global level, these cases reinforce the need for stricter regulation of transplant tourism, standardized donor screening protocols, and improved access to sensitive diagnostic tools such as PCR in transplant centers receiving returning transplant recipients [9-11, 16-18]. Increased clinician awareness and systematic reporting of donor-derived infections are essential to reducing preventable morbidity associated with malaria in solid organ transplant recipients [22, 23].
Conclusion
Malaria remains an important yet under-recognized threat in solid organ transplant recipients, particularly in those undergoing transplant tourism in endemic regions. These two cases demonstrate how immunosuppressive regimens—especially anti-thymocyte globulin, tacrolimus, and mycophenolate mofetil—may attenuate classical febrile responses, leading to delayed diagnosis despite high parasitemia. Atypical or afebrile presentations should not exclude malaria from consideration.
Unexplained anemia, thrombocytopenia, encephalopathy, or graft dysfunction following overseas transplantation should prompt urgent malaria evaluation using peripheral blood smear with or without molecular testing. Early initiation of intravenous artesunate is associated with favorable outcomes, although careful monitoring for delayed hemolysis and drug interactions with calcineurin inhibitors remains essential. Incorporating structured malaria risk assessment into post-transplant follow-up and strengthening donor screening standards in overseas transplant programs are critical steps toward improving patient and graft survival.
Conflict Of Interest: The authors have no conflicts of interest regarding the article, its authorship, or publication.
References
- World Health Organization. (2022) World malaria report 2022. WHO.
- Fikadu M, Ashenafi E (2023) Malaria: an overview. Infection and Drug Resistance. 16: 3339-47.
- White NJ, Pukrittayakamee S, Hien TT, Faiz MA, Mokuolu OA, et al. (2014) Malaria. Lancet. 383(9918): 723-35.
- Kotton CN, Lattes RA (2009) AST Infectious Diseases Community of Practice. Parasitic infections in solid organ transplant recipients. American Journal of Transplantation. 9: S234-51.
- Hung CC, Chang SC, Chen YC, Hsieh WC (1994) Transmission of Plasmodium falciparum malaria by kidney transplantation. Am J Trop Med Hyg. 51(3): 311-4.
- Turkmen A, Sever MS, Ecder T, Erek E, Eraksoy H, et al. (1996) Donor-originated falciparum malaria in renal transplant recipients. Clin Transplant. 10(6 Pt 1): 494-8.
- Bemelman FJ, Senev A, Peeters A, Ijzermans JN, Weimar W (2004) Transmission of Plasmodium falciparum by renal transplantation. Transplantation. 77(3): 478-80.
- Einollahi B, Lessan-Pezeshki M, Pourfarziani V, Nafar M, Firouzan A, et al. (2008) Malaria in kidney transplant recipients: report of four cases. Transplant Proc. 40(1): 203-5.
- Rosso F, Agudelo Rojas OL, Suarez Gil CC, Lopez Vargas JA, Gómez‐Mesa JE, et al. (2021) Transmission of malaria from donors to solid organ transplant recipients: a case report and literature review. Transplant Infectious Disease. 23(4): e13660.
- Chen LH, Wilson ME (2023) The globalization of healthcare: implications of medical tourism for the infectious disease clinician. Clinical infectious diseases. 57(12): 1752-9.
- Soto TM, Kotton CN (2021) Infectious disease complications of transplant tourism. Expert Review of Anti-infective Therapy. 19(6): 671-3.
- Elagali A, Shubayr M, Noureldin E, Alene KA, Elagali A (2024) Spatiotemporal distribution of malaria in the Kingdom of Saudi Arabia. Tropical Medicine and Infectious Disease. 9(1): 16.
- Abdalal SA, Yukich J, Andrinoplous K, Harakeh S, Altwaim SA, et al. (2023) An insight to better understanding cross border malaria in Saudi Arabia. Malaria Journal. 22(1): 37.
- Schofield L, Mueller I (2006) Clinical immunity to malaria. Curr Mol Med. 6(2): 205–221.
- Bousema T, Okell L, Felger I, Drakeley C (2014) Asymptomatic malaria infections: detectability, transmissibility and public health relevance. Nat Rev Microbiol. 12(12): 833–840.
- Tangpukdee N, Duangdee C, Wilairatana P, Krudsood S (2009) Malaria diagnosis: a brief review. Korean J Parasitol. 47(2): 93– 102.
- Snounou G, Viriyakosol S, Zhu XP, Jarra W, Pinheiro L, et al. (1993) High sensitivity of detection of human malaria parasites by the use of nested polymerase chain reaction. Mol Biochem Parasitol. 61(2): 315–320.
- Imwong M, Hanchana S, Malleret B, Rénia L, Day NP, et al. (2014) High-throughput ultrasensitive molecular techniques for quantifying low-density malaria parasitemias. J Clin Microbiol. 52(9): 3303–3309.
- World Health Organization (2015) Guidelines for the treatment of malaria. 3rd ed. WHO.
- Sabé N, González-Costello J, Oriol I, Sánchez-Salado JC, Ortega S, et al. (2014) Donor-derived Plasmodium falciparum malaria in a heart transplant recipient successfully treated with artesunate. Transpl Infect Dis. 16(6): 1008–12.
- Plewes K, Leopold SJ, Kingston HWF, Dondorp AM (2015) Post- artesunate delayed haemolysis after treatment of severe malaria. Malar J. 14: 379.
- Fabiani S, Fortunato S, Bruschi F (2018) Solid organ transplant and parasitic diseases: a review of the clinical cases in the last two decades. Pathogens. 7(3): 65.
- Fishman JA, Grossi PA (2014) Donor-derived infection—the challenge for transplant safety. Nature Reviews Nephrology. 10(11): 663-72.