Almeida GV1*, Brito T2, Katerenchuk V3, Correia M4, Teixeira A1, Ana Isabel Gomes da Silva5
1Serviço de Ginecologia e Obstetrícia.
2Serviço de Pediatria.
3Serviço de Anestesiologia.
4Serviço de Cirurgia Geral Centro Hospitalar de Setúbal, E. P. E.
5Centro Hospitalar de Setúbal, Portugal.
*Corresponding Author: Almeida GV, Serviço de Ginecologia e Obstetrícia,.
Abstract
Abdominal pregnancy is a rare form of ectopic pregnancy with very high morbidity and mortality for both the mother and fetus. Advanced abdominal pregnancy (AAP) could potentially lead to devastating consequences.
Diagnosis and management can pose some difficulties, especially in advanced gestational age. A high index of suspicion is vital in making a prompt diagnosis in such situations. A case of abdominal pregnancy that resulted in a live healthy newborn at a Portuguese hospital is presented.
Introduction
Ectopic pregnancy represents about 1–2% of all pregnancies, with 95% occurring in the fallopian tube. Abdominal pregnancies represent just about 1% of ectopic pregnancies. The incidence of abdominal pregnancy differs in various publications and ranges between 1: 10000 and 1:30000 pregnancies [1].
Primary peritoneal pregnancy is a rare entity whose pathogenesis remains controversial; it was defined as the implantation of the fertilized egg on the pelvic peritoneum. In 1942, Studdiford [2] reported a case of an early ruptured pregnancy on the posterior aspect of the uterus, which, based on its relationship to the interstitial portion of the tube and the peritoneum, was accepted as the first reported primary peritoneal nidation.
Studdiford described the pathogenesis to be related to the ovum expelled from the follicle that becomes fertilized and develops over a period of eight to nine days without being captured by the fimbriated extremity of the tube, which might lead to aberrant nidation in any tissue, regardless of its origin, with which the blastocyst is in contact. Except for the germinal epithelium of the ovary, such tissue must be either visceral or parietal peritoneum. Such a pregnancy must be termed a primary peritoneal pregnancy.
After that, primary peritoneal pregnancy was clinically distinguished from secondary peritoneal pregnancy by Studdiford's established criteria. Studdiford's measures include the following:
- Normal tubes and ovaries with no evidence of recent or remote injury,
- An absence of any evidence of a retroperitoneal fistula,
- The presence of a pregnancy-related exclusively to the peritoneal surface and early enough to eliminate the possibility of secondary implantation following a primary nidation in the tube.
Abdominal pregnancy has remained a significant challenge worldwide, especially in developing countries where there are limitations in diagnostic resources. The most important approach is to be vigilant for the unexpected, as most patients present with no specific symptoms or clinical signs.
Maternal mortality and morbidity are also very high, especially if the condition is not diagnosed and managed appropriately. These pregnancies generally do not get to term and usually result in the extraction of a dead fetus. Maternal mortality is 0.5– 18%, and the perinatal mortality rate is 40–95%. [3,4]
Abdominal pregnancy at term with a healthy viable fetus is, therefore, a scarce condition, and very few such cases have been published during the last ten years. We present a case of abdominal pregnancy that resulted in a term live baby without malformations.
Case Report
A pregnant 25-year-old Gesta 0 Pakistani female presented to our health facility complaining of pelvic pain and distension, nausea, and vomiting at an estimated gestational age of 30 weeks and 6 days. Past medical history was significant for anemia, without adequate study, meaning the etiology was unknown. A vital language barrier was noted, which prevented us from obtaining a detailed history.
The patient had two previous obstetrical ultrasounds in Pakistan, carried out at 19 and 26 weeks, with no particularities described.
On physical examination, she was severely pale; her pulse rate was 116-135 beats/min, and her blood pressure was 90/60 mmHg. Her abdomen was hypertonic and asymmetrically enlarged, with a uterine height lower than the reported gestational age. The fetal heart rate was 105 beats/min at admission. A vaginal examination revealed that the cervical os was closed and the pouch of Douglas was full. There were no signs of vaginal bleeding.
Emergent abdominal ultrasound was performed to confirm fetal heart rate, and the fetus was in an oblique position in the maternal abdomen, with the head in the left iliac fossa. An anterior placental implantation site and severe oligoamnios were noted. Ultrasound confirmed a fetal heart rate of 100 beats/min.
After fluid resuscitation, the patient became hemodynamically stable and had an initial hemoglobin level of 10.8 g/dL, with a leucocytosis of 19 300 and 84% neutrophils. Serologic testing for sexually transmitted diseases was negative.
After one hour, her pain worsened, and she became hypotensive and had maintained tachycardia (124-138 pulses/minute). Concomitantly, the fetus presented sustained bradycardia. The decision was made to perform an emergency cesarean. At this moment, the first diagnostic hypothesis was a uterine rupture.
The patient had an emergency exploratory laparotomy. A general surgeon and a neonatologist were present at the surgery. Intraoperative findings were as follows:
- hemoperitoneum of about 1,2 L with clotted blood of almost 500 g;
- anterior placenta that was firmly attached to the posterior surface of the uterus, omentum, and the left infundibulopelvic ligament;
- no amniotic fluid and a confirmed rupture of membranes;
- a live female baby located in the Pouch of Douglas (POD), weighing 1820gr with APGAR scores of 5 at 1 minute and 8 at 5 minutes;
- both right and left ovaries were normal, and the bulky uterus corresponded to about 15 weeks' size;
- iatrogenic laceration of the bladder caused by extraction of the fetus.
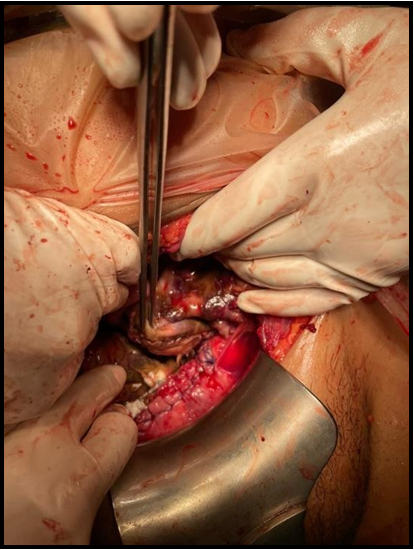
The placenta was dissected away safely from the gastrocolic omentum, the left sidewall of the pelvic cavity, and the abdominal sidewall, but it was difficult to separate from the uterus. Massive bleeding was seen after placenta removal. To avoid uncontrolled bleeding, a total abdominal hysterectomy was conducted, followed by bladder repair [Figure 1]. Both ovaries were preserved.
Figure 1: Placental implantation site and sutured bladder.
There was an area of maintained bleeding tissue in the mesosalpinx and omentum, which was initially controlled with direct compression. After the histerectomy, the thick band connecting the placenta, the omentum and infundibulopelvic ligament was clamped with electrosurgical generator LigaSure®. The placenta was wholly delivered without any remnant left in situ. Abdominal drains were inserted into theleft paracolic gutter and the pouch of Douglas. The remaining bleeding areas were secured by cauterization and application of topical hemostatic agents.
Both the placenta and the tissue attached to it were removed and sent for histopathology. Pathologic findings of excised masses from the initial operation were:
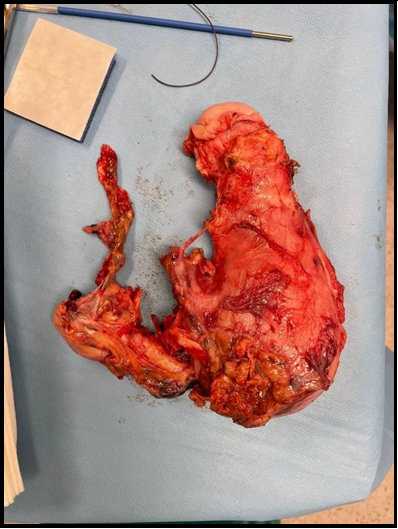
1. Uterus: presence of decidualized endometrium, as a result of extra- uterine pregnancy. Placental implantation site is defined in the uterus serosa and adherent para-uterine tissues. [Figure 2].
2. Placenta: degenerated villous trophoblasts, consistent with a third trimester placenta, with focal edema (hydrops) and denatured areas of varying size and shape and pathological trophoblast cell proliferation.
Figure 2: Augmented sized uterus and placenta implanted on fundical and posterior surface of the uterus.
The cesarean was performed under balanced general anesthesia after a rapid sequence induction. The massive transfusion protocol was activated shortly after the extraction of the baby due to severe blood loss, causing hemodynamic instability. During the intra-operative period, the patient crashed, and pulseless electrical activity was perceived. Immediate cardiopulmonary resuscitation was initiated, and 1 mg of epinephrine was administered. Return of spontaneous circulation ensued within two minutes. Patient stabilization was possible after a transfusion of four red blood cells and one unit of fresh frozen plasma. After the surgery, the patient was transferred to the intensive care unit, where she was intubated within the first 24 hours. Posttransfusion hemoglobin was 7,4 g/dL, hematocrit 21,7%. The fetus was delivered and showed no structural abnormality. The newborn was transferred to a neonatal intensive care unit with mechanical ventilation. She was weaned to noninvasive ventilation on the second day of life and was transferred back to her local hospital after four days without ventilatory support. She needed phototherapy for three days and had a good recovery.
The postoperative phase was uneventful. The abdominal drains were removed on the 5th postoperative day, and bladder catheterization was maintained for 21 days.
On the 21st postoperative day, the patient was discharged uneventfully from the hospital with a healthy baby.
Discussion
The clinical presentation of an abdominal pregnancy can differ from that of a tubal pregnancy. Although there may be significant variability in symptoms, severe lower abdominal pain is one of the most consistent findings. [5] Also, persistent abdominal pain, painful fetal movements, weight loss, abnormal presentations, uneffaced cervix, vaginal bleeding, and palpation of an abdominal mass distinct from the uterus are features that may arouse suspicion.
However, the risk factors for abdominal pregnancy are the same as those for tubal pregnancy. [6] The recent use of intrauterine devices, a history of surgery, pelvic inflammatory disease, and a history of sexually transmitted disease are associated with an increased risk of ectopic pregnancy. This patient had not been using any contraception and did not report any history of other risk factors.
We presented a case of abdominal pregnancy with the delivery of a viable baby. This patient presented with amenorrhea, abdominal pain, and general malaise, which suggested a maternal or obstetrical complication; however, the diagnostic workup was not noticeable. As shown in this case, an ultrasound scan may not accurately diagnose late gestational abdominal pregnancy, mainly when this hypothesis is not posed. Magnetic resonance imaging and computed tomography scan may be indicated to diagnose late abdominal pregnancies.
This case may be considered a secondary abdominal pregnancy since the presentation did not meet Studdiford's criteria. Although the diagnosis was made intraoperatively, there was no evidence of uteroperitoneal fistula, and both the ovaries and tubes were expected; the diagnosis was not made in an early phase, which cannot exclude the possibility of secondary implantation following a primary nidation in the box.
Abdominal pregnancy is often associated with congenital malformations in about 40% of cases, and only 50% of these babies survive up to one-week post-delivery. [7,8] However, in this case, the neonate had no structural deformities.
The management of abdominal pregnancy depends on the estimated gestational age at presentation and the clinical presentation. Previable gestations require immediate laparotomy, irrespective of the clinical status. However, for asymptomatic viable pregnancy, hospital-based conservative management is advocated till lung maturity is achieved. As presented in this case, symptoms such as vaginal bleeding, generalized abdominal pain, evidence of hemoperitoneum, and worsening vital signs require urgent operative intervention.
The most severe complication of abdominal pregnancy is bleeding from the placental site. Decisions such as clamping the cord close to the placenta and administration of methotrexate to hasten autolysis and degeneration of the placenta.
It may sometimes be necessary. It is generally recommended to leave the placenta in situ and make a follow-up with human chorionic gonadotropin levels.[9]
In our case, the placenta was mainly attached to the posterior surface of the uterus and omentum and was irretrievably removed, at least in part, during the extraction of the fetus due to its anterior location. This conservative approach may, however, be complicated by intraperitoneal infection and adhesions.
Despite the excellent outcome of this case, it is essential to appreciate that it is decisive to have a correct and early diagnosis since it allows time for thoughtful preparation of the patient, family, and medical team. However, to be diagnosed, abdominal pregnancy must first be considered and requires a high index of suspicion, which did not happen in this situation, probably due to advanced gestational age.
Conclusion
Early diagnosis of peritoneal pregnancy is challenging but is essential due to the life-threatening nature of the condition. We reported a case of abdominal pregnancy that was successfully treated despite incidental diagnosis. This case highlights the atypical presenting features of advanced abdominal pregnancy and the need to consider this diagnosis. It is also aimed at showing the difficulty of diagnosis of advanced abdominal pregnancy in a resource environment.
*No potential conflict of interest relevant to this article was reported.
References
- Nwobodo EI (2004) Abdominal pregnancy. A case report. Ann Afr Med. 3(4): 194–196.
- Studdiford WE (1942) Primary peritoneal pregnancy. Am J Obstet Gynecol. 44(3): 487–491.
- Kassam M. Abdominal pain in pregnancy. In: David KJ, Phillips JS, Carl PW, Bernard G, editors. High risk pregnancy management option, 2. London: WB Saunders; 2007. p. 996–7.
- Martin JN Jr, McCaul JF (1990) Emergent management of abdominal pregnancy. Clin Obstet Gynecol. 33(3): 438–447.
- Nkusu Nunyalulendho D, Einterz EM (2008) Advanced abdominal pregnancy: case report and review of 163 casesreportedsince 1946. Rural Remote Health. 8(4): 1087.
- Berek JS. Berek & Novak’s Gynecology. 14th ed. Philadelphia: Lippincott Williams & Wilkins; 2007. p. 628.
- Baffoe P, Fofie C, Gandau BN (2011) Term abdominal pregnancy with healthy newborn: A case report. Ghana Med J. 45(2): 81–83.
- Stevens CA (1993) Malformations and deformations in abdominal pregnancy. Am J Med Genet. 47(8): 1189–1195.
- Jianping Z, Fen L, Qiu S (2008) Full-Term Abdominal Pregnancy. A Case Report and Review of the Literature. Gynecol Obstet Invest. 65(2): 139–141.