Ye Guo MD1,2, Shuchun Wang MD1,2, Ning Wang MD1,2, Xiaojuan chen MD1,2*
1Division of Pediatric Blood Diseases Center, State Key Laboratory of Experimental Hematology, National Clinical Research Center for Blood Diseases, Haihe Laboratory of Cell Ecosystem, Institute of Hematology & Blood Diseases Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Tianjin.
2Tianjin Institutes of Health Science, Tianjin 301600, China.
*Corresponding Author: Xiaojuan Chen MD, Division of Pediatric Blood Diseases Center, State Key Laboratory of Experimental Hematology, National Clinical Research Center for Blood Diseases, Haihe Laboratory of Cell Ecosystem, Institute of Hematology & Blood Diseases Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Tianjin and Tianjin Institutes of Health Science, Tianjin 301600, China.
Abstract
A rare case of isolated intracranial myeloid sarcoma (MS) with PML: RARα fusion was identified. Histopathological examination of surgically resected brain tissue demonstrated a malignant neoplasm with limited lineage specificity. DNA-based next-generation sequencing detected the PML:RARα fusion, which enabled definitive diagnosis of isolated intracranial MS with this molecular alteration. Surgical resection followed by systemic therapy guided by acute promyelocytic leukemia (APL) protocols achieved durable remission in this patient. A rare case of isolated intracranial myeloid sarcoma (MS) with PML:RARα fusion was identified. Histopathological examination of surgically resected brain tissue demonstrated a malignant neoplasm with limited lineage specificity. DNA-based next-generation sequencing detected the PML:RARα fusion, which enabled definitive diagnosis of isolated intracranial MS with this molecular alteration. Surgical resection followed by systemic therapy guided by acute promyelocytic leukemia (APL) protocols achieved durable remission in this patient.
Introduction
Myeloid sarcoma (MS) is an extramedullary malignant neoplasm composed of myeloid blasts or immature myeloid cells and may arise at virtually any anatomical site [1]. Isolated MS is defined by the absence of prior or concurrent leukemia and by negative bone marrow findings at diagnosis, with an estimated annual incidence of approximately 0.7 cases per million children [2]. Accurate diagnosis of isolated MS remains challenging and depends on confirmation of myeloid lineage in tumor cells [2]. A rare case of isolated intracranial MS in a 15-year-old female with PML:RARα fusion is presented, in which remission was achieved following treatment guided by the Chinese Children’s Cancer Group Acute Promyelocytic Leukemia 2017 (CCCG-APL-2017) protocol.
Case Report
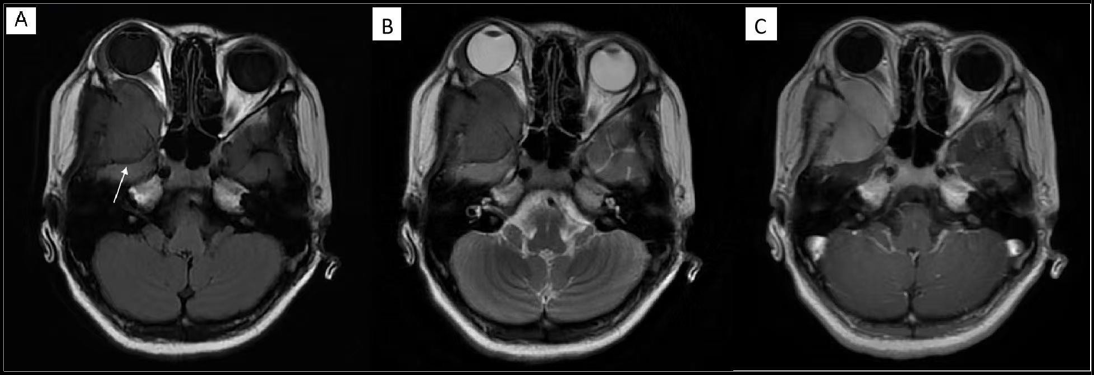
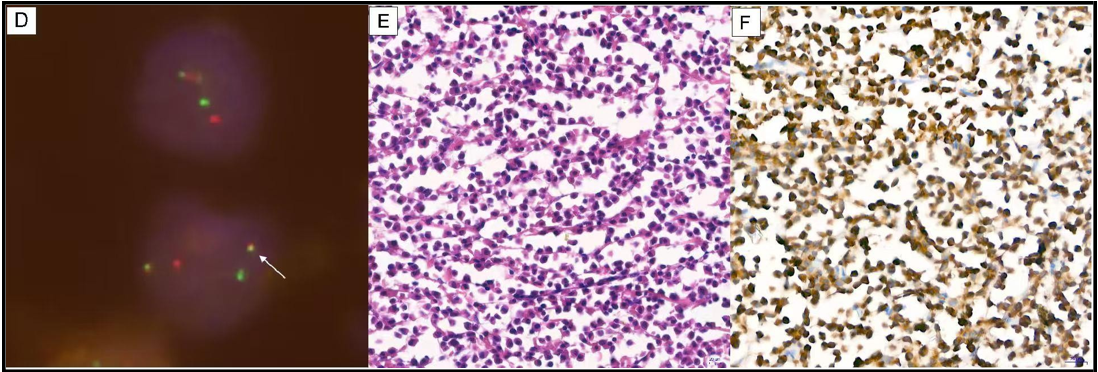
A rare case of isolated intracranial MS with PML:RARα fusion was identified in a 15-year-old female who presented with a 2-week history of persistent headache and swelling of the right cheek. Magnetic resonance imaging revealed a 4.7 × 4.6 × 4.8 cm mass in the right temporal region (Figure 1). Neuronavigation-guided craniotomy was performed to achieve surgical resection. Histopathological evaluation demonstrated a small, round-cell malignant neoplasm with areas of necrosis. Immunohistochemical (IHC) staining of core biopsy material showed that immature myeloid cells were positive for myeloperoxidase and Ki-67 (15%) and negative for CD34, CD30, CD68, CD138, CD20, and CD163. DNA-based targeted next-generation sequencing of the resected tissue confirmed the presence of PML:RARα fusion with a positive rate of 44.57%. Fluorescence in situ hybridization demonstrated 82% positivity for the same fusion (Figure 2). Peripheral blood counts, coagulation parameters, bone marrow examination, and cerebrospinal fluid (CSF) analysis were unremarkable, establishing the diagnosis of isolated intracranial MS with PML:RARα fusion.
Figure 1: Magnetic Resonance Imaging (MRI) Of The Brain.
A: T1-weighted MRI showing an isointense lesion (mass) in the right middle cranial fossa, involving adjacent meninges and temporal bone (white arrow).
B: T2-weighted MRI showing low signal intensity of the lesion.
C: Contrast-enhanced MRI showing marked enhancement of the lesion (mass).
Figure 2: Histopathological and molecular characterization of brain tissue.
D: Fluorescence in situ hybridization analysis of resected brain tissue demonstrating positivity for the PML:RARα fusion gene (white arrow).
E: Hematoxylin and eosin (H&E) staining (×40) showing diffuse infiltration of abnormal promyelocytes with moderate cell size, abundant eosinophilic cytoplasm, and folded nuclei in a subset of cells.
F: Myeloperoxidase (MPO) staining (×40) demonstrating MPO-positive tumor cells.
Postoperatively, headache symptoms improved; however, swelling of the right temporal region, diplopia caused by impaired abduction of the right eye, and numbness of the right facial area persisted for several months. Treatment following the CCCG-APL-2017 protocol was initiated [3]. Radiotherapy was not administered due to the complete surgical resection of the tumor. Three courses of intrathecal triplet therapy consisting of methotrexate, cytarabine, and dexamethasone were administered. CSF cytology, protein analysis, and flow immunophenotyping remained normal. After one cycle of chemotherapy with arsenic trioxide (ATO) and all-trans-retinoic acid (ATRA), swelling and sensory symptoms resolved gradually, and complete molecular remission was maintained at 14-month follow- up.
Discussion
This case was exceptional in that isolated intracranial MS with PML:RARα fusion was identified in a pediatric patient, a presentation not previously reported in the published literature (PubMed). Acute myeloid leukemia (AML) presenting as isolated MS is rare, accounting for approximately 1% of cases [4]. In acute promyelocytic leukemia (APL), MS occurs infrequently and most often at relapse, whereas presentation as isolated extramedullary disease without bone marrow involvement is exceedingly uncommon [5]. The PML:RARα fusion is typically associated with APL, an aggressive leukemia subtype for which durable remission is achieved in 98% of patients treated with ATRA/ATO therapy [6, 8]. In contrast, isolated intracranial MS with PML:RARα fusion represents an exceptionally rare clinical scenario, and histopathology alone lacks sufficient specificity for definitive diagnosis. Although H&E staining may suggest promyelocytic morphology, morphological features alone are inadequate to establish the diagnosis in the absence of molecular confirmation. Molecular profiling using next-generation sequencing and related assays therefore plays a critical role in accurate classification of this disease. The favorable therapeutic response observed in this case supports the concept that MS with PML:RARα fusion shares core differentiation and apoptosis mechanisms with APL and responds effectively to ATRA/ATO-based therapy [7]. This outcome provides a rational treatment reference for future patients presenting with similar clinicopathological features.
Conclusion
Isolated MS typically progresses to systemic disease within three to six months in the absence of treatment [9, 11]. This case underscores the importance of combining surgical resection with systemic therapy guided by APL protocols to achieve durable remission and offers a therapeutic framework for similar future cases.
Acknowledgements: The authors would like to thank all participants for their contributions to the study.
Conflicts Of Interest: The authors of this manuscript declare no potential conflicts of interest.
Ethics Consideration
This study was approved by the IRB of the Institute of Hematology & Blood Diseases Hospital, Chinese Academy of Medical Sciences (Approval No. IIT2016008-EC-3). Written informed consent-to- disclose was obtained from the patient.
Funding: This work was supported by a grant from the National Natural Science Foundation of China (82070201 [Y.G.]).
Author Contributions
Ye Guo: Contributed to study design and drafting of the article.
Shuchun Wang and Ning Wang: Contributed to patient management and drafting of the article.
Xiaojuan Chen: Contributed to study design, drafting of the article, and data analysis and interpretation.
References
- Lontos K, Yabes JG, Farah RJ, Boyiadzis M (2021) Impact of upfront chemotherapy on overall survival in isolated myeloid sarcoma. Leukemia. 35(4): 1193-1196.
- Antic D, Elezovic I, Milic N, Suvajdzic N, Vidovic A, et al. (2013) Is there a "gold" standard treatment for patients with isolated myeloid sarcoma?. Biomed Pharmacother. 67(1): 72-7.
- Chang L, Gao J, Lei X, He Y, Zhuang S, et al. (2025) A multicenter single-arm clinical study of Chinese children's cancer group-acute promyelocytic leukemia-2017 (CCCG-APL-2017) protocol. Signal Transduct Target Ther. 22; 10(1): 267.
- Pileri SA, Ascani S, Cox MC, Campidelli C, Bacci F, et al. (2007) Myeloid sarcoma: clinico-pathologic, phenotypic and cytogenetic analysis of 92 adult patients. Leukemia. 21(2): 340-350.
- Du Y, Yang K, Ling Y, Zhang Y, Gong Y (2025) A case report of acute promyelocytic leukemia with myeloid sarcoma of the lumbar spine and literature review. Front Med(Lausanne). 11: 1507716.
- Yilmaz M, Kantarjian H, Ravandi F (2021) Acute promyelocytic leukemia current treatment algorithms. Blood Cancer J. 11(6): 123.
- Tomita A, Kiyoi H, Naoe T (2013) Mechanisms of action and resistance to all-trans retinoic acid (ATRA) and arsenic trioxide (As2O3) in acute promyelocytic leukemia. Int J Hematol. 97(6): 717-725.
- Bercier P, de Thé H (2024) History of Developing Acute Promyelocytic Leukemia Treatment and Role of Promyelocytic Leukemia Bodies. Cancers. 16(7): 1351.
- Yamashita T, Nishijima A, Noguchi Y, Narukawa K, Oshikawa G, et al. (2019) Acute promyelocytic leukemia presenting as recurrent spinal myeloid sarcomas 3 years before developing leukemia: A case report with review of literature. Clin Case Rep. 8; 7(2): 316-321.
- Gu Y, Zheng H, Mo S, Guo T, Chen L, et al. (2023) De novo myeloid sarcoma mimicking gynecological tumors: a retrospective case series of eight patients. BMC Womens Health. 29; 23(1): 141.
- Begna KH, Kittur J, Yui J, Gangat N, Patnaik MM, et al. (2021) De novo isolated myeloid sarcoma: comparative analysis of survival in 19 consecutive cases. Br J Haematol. 195(3): 413-416.